Blogs
Malika Menoud – Installation of a CF-IRMS and methane extraction system
Blog written by Malika Menoud
Installation of a Continuous Flow Isotope Ratio Mass Spectrometer (CF-IRMS or for us just mass spec) and methane (CH4) extraction system for continuous isotopic measurements in ambient air during 17th to 26th of May 2018, AGH University of Science and Technology, Krakow, Poland
First: Time to explain what this “system” does!
The idea is to measure methane (CH4) in ambient air. It is present as a trace gas, with concentrations around 2 ppm, but its high greenhouse potential makes it of high relevance for climate change studies.
For this we need to separate the methane from the other air compounds, by freezing it at very low temperature (below -135 °C). This first trap is the first step of the extraction. The amount of methane resulting from this is very small (around 0.1 µl), as it is extracted from only 40 or 60 ml of air. It needs now to be concentrated for the instrument to be able to detect it further on. Another trap at very low temperature is used for this step of so called “focusing”. The cooling of both traps is performed with a gas compressor.
The methane needs then to be converted on either CO2 or H2, for carbon 13 (13C) or deuterium (2H) isotopes measurements respectively. This is done by combustion at 1150 °C with the injection of pure O2 in one oven, and by pyrolysis at 1375 °C in another oven. The resulting CO2 or H2 goes through a GC column to stay well-separated. The CO2 will be also dried to make sure no residual water will affect the measurements. Now the sample (or what’s left from it) can be injected in the IRMS for the determination of CH4 isotopic ratios.
In the IRMS (isotopic ratio mass spectrometer), the molecules will be ionized, which means they will get a charge. Then they will travel in a magnetic field, in which the different masses will follow a different trajectory. At the end, some detectors will catch the molecules at different locations, to quantify their different masses. Because of the small mass difference, this allows us to distinguish between isotopes of the same molecule.
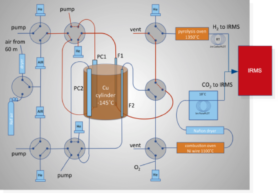 The whole system is illustrated in the scheme. We see two parallel lines for methane extraction, one will be for 13C measurements, and the other for 2H. They are happening alternatively as the IRMS can measure one sample at a time. Time-wise, one measurement is taking 30 min in total, including 10 min of IRMS acquisition. While a sample is measured, another one is being extracted. Thus we’re able to provide one value every 20 min.
The whole system is illustrated in the scheme. We see two parallel lines for methane extraction, one will be for 13C measurements, and the other for 2H. They are happening alternatively as the IRMS can measure one sample at a time. Time-wise, one measurement is taking 30 min in total, including 10 min of IRMS acquisition. While a sample is measured, another one is being extracted. Thus we’re able to provide one value every 20 min.
|
Installation of the system 1st day: 17th of May 2018 |
|
 |
1) The boxes arrived yesterday, after being transported all the way from Utrecht, the Netherlands. They are lying in the corridor, waiting to be opened. |
 |
2) The mass spec arrived safely, let’s hope nothing is damaged inside. |
 |
3) First, we’ve made some space in the lab for the system to be installed. The desk on the right is placed for the computers to sit. Some cylinders are already here. We will need pure helium, CO2 and H2, as well as two working standard that we’re bringing with us. |
 |
4) The mass spec is plugged in and switched on. The analyzer pump that sits inside is the first thing to start, as it can take some time. The two computers are now connected to the network. |
 |
5) The GasBench is the unit from where the sample or reference gas are introduced to the mass spec. It is placed on a self-made shelf fixed on top of the mass spec. Here will also sit the cooler for the gas chromatography (GC) column. |
 |
6) The inlet capillary is connected on top of the IRMS source, where the sample will first enter. We can now flush it with helium, that is the carrier gas used throughout the whole system. |
 |
7) Placing regulators and opening cylinders is a fun group activity! From left to right, Jarek, Mila and Carina. |
|
2nd day: 18th of May 2018 |
|
 |
8) The next day, we’re taking the extraction system out of its box. The traps and ovens are all placed on the same rack, with the compressor, pumps and power box below. The rack will sit next to the mass spec. |
 |
9) The two traps are mounted on a copper block, which is on the cold end of the compressor. We connected the temperature sensors of each trap on the electronic board before covering the unit under a vacuumed and thermally insulated chamber. |
 |
10) The valve on the front allows to connect 6 samples for measurements, in addition to the ambient air pumped from the roof. This and the other valves work with compressed air. On the right sit 8 mass flow controllers (MFC), that regulate the flow rate through the different parts of the system. |
 |
11) The two ovens (combustion and pyrolysis) are placed in the back. A new ceramic tube is placed in each of them, for the gas to go through. After concentration, the methane is flowing in 36 µm capillaries. These are connected to the ovens, and from there to the GC columns and GasBench. The system is now closed. |
 |
12) On the bottom of the rack is a forest of cables that were untied to make the connection with the computers. On the screen of the Labview computer, we can see if the MFC are measuring well by adjusting the helium flow and check how they respond. Helium is now flowing through the system! |
 |
13) Copernicus, our local mascot who sits here to guard the IRMS. |
 |
14) One of the two GC columns need to be cooled. Carina is mounting it in a new structure that can be placed in the new cooling chamber. But this will be tested tomorrow as now is time to get some rest. |
|
3rd day: 19th of May 2018 |
|
 |
15) The GC is now in its temperature regulated box. It will stay next to the GasBench on top of the mass spec. The column needs to be heated first to get rid of the water, then we’ll cool it down to around 0°C. |
 |
16) The system is ready to be tested. Let’s switch the compressor on! After causing one or two power break, it eventually started. Now the traps are being cooled down. |
 |
17) The Isodat computer is connected to the GasBench and IRMS. We start by checking the argon signal. Argon is naturally present in air, and indicates weather there is a leak. Afterwards we’re checking the signal of the two reference gas: pure CO2 and pure H2. We use these tests to optimize the IRMS settings for the measurements of these compounds. We can also identify some problems that we can fix prior to measure our first air sample. |
 |
18) There are always unexpected issues that need to be solved. A bending capillary at the open-split affects the flow in the IRMS. More space was made in the open-split by replacing a capillary with a thiner one. Problem solved! Another issue arises from a USB connection that was not working, which prevents the two computers to communicate with each other. After several days trying different solutions, this connection was established. |
 |
19) What we want to measure is the air from outside, at a relatively elevated point. An inlet tube is attached to a 20 m mast sitting on the roof. The tube crosses the building until the lab (black thick tube from top to bottom). It is connected to a powerful pump, down after the valve. A smaller tube is diverted from the main line (transparent tube on the right). We’ve placed a pressure sensor at this crossing, and a temperature sensor nearby (the two gray cables). These measurements are visible in real time on the Labview computer. |
 |
20) The roof air line is split into the system’s inlet and the Los Gatos (LGR) inlet. The LGR is an instrument that measures CH4mixing ratio in atmospheric air, at a frequency of 1 Hz, and a precision of 1 ppb. We can see it on the left of the desk, with a small monitor on top. In this experiment, we will use it to compare with the data provided by the IRMS, to eventually correct it. After the last adjustments of some extraction parameters (waiting times, temperatures, …) to optimize the peak shapes, it is ready to run! In a few months we will uninstall everything, collect the data and bring the material back to Utrecht! I’m sure it will give exciting results … to be followed! |
21) Waiting for the first sample peak to appear…
You can also enjoy the warbling sound of the compressor, singing together with all the pumps.

